Technical topics and roadmap
Interop Roadmap 2024-2025
Dynamic roadmap
Archive: Interop Roadmap 2023-2024
The roadmap is a document that is subject to continuous development and annual revision. The roadmap is not a national strategy, but a work programme that focuses on pragmatic feasibility.
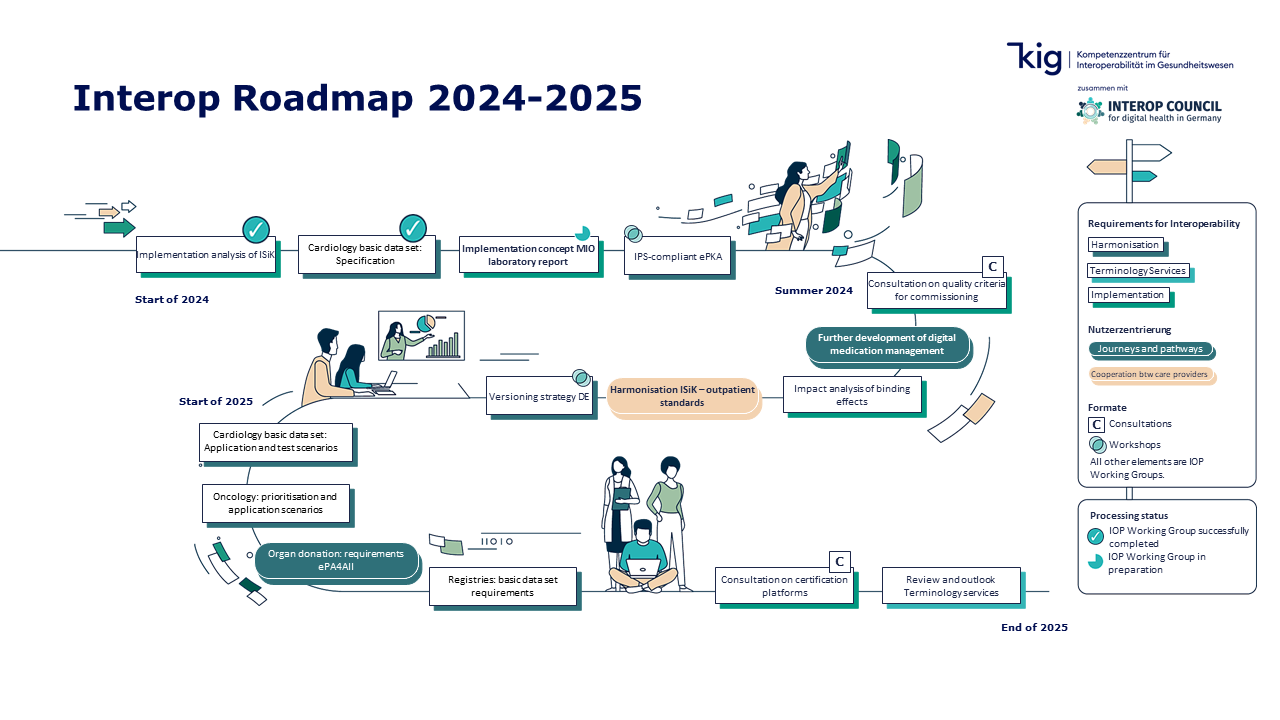
Roadmap
The topics in detail
Intersectoral co-operation
Intersectoral co-operation
| Specialist user journey medication process | Description/objective: To visualise the medication process holistically and across sectors, taking into account international standards
Benefit/added value: Increase in drug therapy safety, cross-sector data exchange
Operationalisation/implementation: Working group, development of recommendations for action for harmonisation or recommendation of certain standards
Required experts / collaboration: HL7, MIO42, ePA, e-prescription, pharmacists (also inpatient) Timeframe: HY2 2023 |
| Intersectoral board for rare diseases | Description/goal: -
Benefit/added value: Cross-practitioner analysis of data to identify suspected cases of rare diseases and support their treatment, similar to tumour boards
Operationalisation/implementation: Working group, development of cross-sector journey
Required experts / collaboration: Outpatient + inpatient doctors + ? Period: HY 2 2024 |
Journeys & Pathways
Journeys & Pathways
| Oncological specialist user pathway | Description/goal: Specialist user journey for service providers in oncology (indication tbd)
Benefit/added value: Identification of harmonisation potential in oncology from the perspective of service providers
Operationalisation/implementation: Working group, recommendations for action
Required experts / collaboration: Oncology outpatient & inpatient service providers Period: HY 2 2022 |
| Care Journey | Description/goal: Development of a journey for users from the areas of home care, social care centres and discharge management
Benefit/added value: Identification of interoperability gaps and necessary standardisation tasks.
Operationalisation/implementation: Recommendations for standardisation activities in the care sector
Required experts / collaboration: Nursing staff / nursing experts, nursing IT manufacturers Timeframe: 4 months from March 2023 |
| Collection + evaluation of outpatient use cases | Description/objective: Identification and evaluation of use cases in outpatient care that would gain significant added value through interoperability
Benefit/added value: Based on this, strategies can be developed on how interoperability can be implemented more strongly in outpatient care.
Operationalisation/implementation: Working group, recommendations for action
Required experts / collaboration: outpatient service providers, Mio42/ KBV, gematik Period: HY1 2024 |
| Citizen pathway diabetes diary | Description/objective: Development of a citizen pathway using the example of a diabetes diary
Benefit/added value: Identification of the added value of interoperability for citizens
Operationalisation/implementation: Working group, recommendations for action
Required experts / collaboration: gematik, Mio42, DDG/ diabetes experts Timeframe: HY 2 2024 |
Terminology services
Terminology services
| Terminology server/services | Description/goal: Development of criteria for infrastructure, access, maintenance, services for central terminology servers together with BfArM and gematik
Benefit/added value: Ease of use, better dissemination of international terminologies in DE
Operationalisation/implementation: Recommendations for action for BfArM and gematik
Required experts / collaboration: BfArM, gematik, terminology experts, users and developers
Timeframe: November 2022 - February 2023 |
| Define value set authority | Description/goal: Development of governance for value set providers under the aegis of a value set authority
Benefit/added value: Clear rules for setting up and maintaining coding systems and value lists
Operationalisation/implementation: working group, recommendations for action
Required experts / collaboration: value set provider, BfArM, BMG
Timeframe: HY2 2023 |
Implementation
Implementation
| Validation/ test environments | Referenzvalidator | Description/objective: Based on the reference validator provided by gematik, the aim is to work out how and for what purposes future test modules will be made available on a binding basis
Benefit/added value: Test modules can be used to evaluate the correct implementation of applications.
Operationalisation/implementation: Working group, recommendations for action
Required experts/collaboration: Interface development, standardisation, FHIR, primary system development
Timeframe: November 2022 - February 2023 |
| Test environments | Description/Objective: Development of criteria for the design of test environments for testing new (TI) applicationsBenefit/added value: There is currently no possibility to test new applications in various primary systems in advance.
Benefit/added value: There is currently no possibility to test new applications in various primary systems in advance.
Operationalisation/implementation: Working group, recommendations for action
equired experts/cooperation: Primary system manufacturers, specifiers
Timeframe: HY1 2023 | |
| Implementation of TI applications in primary systems | Description/objective: The realisation of TI applications in primary systems is stalling. Various possible solutions (confirmation procedures, control options in productive operation) are to be discussed.
Benefit/added value: Identify acceleration potential for the implementation of TI applications.
Operationalisation/implementation: Working group, recommendations for action
Collaboration/experts required: Gematik, primary system manufacturers, providers of TI applications
Timeframe: HY2 202 | |
| Binding nature of the interoperable interfaces in accordance with §371 and §373 | Liability ISiK | Description/objective: Investigation of the current status of the binding nature of ISiK. Identification of acceleration potential
Benefit/added value: Accelerate and strengthen the use of standards in the stationary sector..
Operationalisation/ implementation: working group, recommendations for action
Required experts/cooperation: primary system manufacturers, specialised users, specifiers
Period: HY2 2023 |
| Liability AWST | Description/objective: To analyse the current status of the liability of AWST.
Benefit/added value: Accelerate and strengthen the use of standards in the outpatient sector.
Operationalisation/implementation: working group, recommendations for action
Required experts/cooperation: KBV, primary system manufacturers, specifiers
Period: HY1 2023 | |
Harmonisation
Harmonisation
| Interoperable device interfaces | Description/goal: Development of a FHIR-based device interface
Benefit/added value: Replacement of proprietary solutions from the outpatient sector, internationally reusable interface for industry
Operationalisation/implementation: Development of criteria for FHIR interface, if possible commissioning of service provider for development
Required experts/cooperation: device manufacturer, PVS manufacturer
Timeframe: 3 months from February 2023 |
| KIM/ TIM: standardised (structured) data model | Description/objective: Development of criteria and requirements for a standardised data model as a construction kit for data objects in KIM and TIM
Benefit/added value: Dispatch of data in KIM/ TIM (incl. standardisation and structuring).
Operationalisation/implementation: Working group, recommendations for action
Required experts/cooperation: KBV, gematik, manufacturers of TI applications and primary systems, specialist users
Timeframe: HY2 2023 |
| Harmonisation of cardiological standard | Description/objective: Implementation of the working group's recommendations for action (analysis of the status quo of the "heart failure patient journey".
Benefit/added value: still to be defined
Operationalisation/implementation: still to be defined
Required experts/cooperation: still to be defined
Timeframe: HY2 2024 |
Strategies 2030 & Governance
Strategies 2030 & Governance
| Strategy 2030 & Governance | Description/goal: Continuous development of an IOP strategy and IOP governance for Germany
Benefit/added value: Establishment of clear objectives and rules for cooperation between IOP stakeholders in the German healthcare system.
Operationalisation/implementation: still to be defined
Required experts/cooperation: still to be defined
Timeframe: continuous 2023 - 2024 |